During their development tumors acquire the capability to escape immune control. Immune escape is a fundamental trait of cancer, but until recently there was little understanding of how it develops. IDO is an enzyme that we and others have found to drive immune escape in tumors, where IDO is often inappropriately switched on.
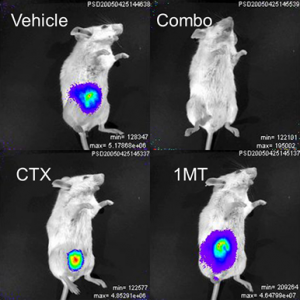
In the present project, we are moving forward to develop our discovery that drug-like small molecule inhibitors of the IDO enzyme stimulate the immune system to attack tumors in an animal in a manner that synergistically leverages the responses that can be achieved with chemotherapy or radiotherapy. The combination of a traditional chemotherapeutic agent, cytoxan (CTX), and 1-methyl-tryptophan (1-MT), a known IDO inhibitor, have demonstrated considerable therapeutic potential (see Combo in Figure 1; tumor is imaged with bioluminiscence).
Figure 1: Effects of combination therapy on mice tumors
 Over the past several years, we have systematically explored and optimized three structural classes of IDO inhibitors through traditional structure-activity relationship studies. These include the dithiocarbamates (Figure 2 and J. Med. Chem. 2006, 49(2), 684-692), the naphthoquinones (Figure 3 and J. Med. Chem. 2008, 51(6), 1706-1718) and the phenyl-imidazoles (Figure 4 and J. Med. Chem. 2008 51(16), 4968-4977).
Over the past several years, we have systematically explored and optimized three structural classes of IDO inhibitors through traditional structure-activity relationship studies. These include the dithiocarbamates (Figure 2 and J. Med. Chem. 2006, 49(2), 684-692), the naphthoquinones (Figure 3 and J. Med. Chem. 2008, 51(6), 1706-1718) and the phenyl-imidazoles (Figure 4 and J. Med. Chem. 2008 51(16), 4968-4977).
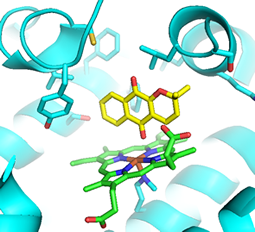
Figure 2: Dithiocarbamate structure-activity relationship study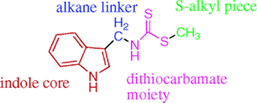 Figure 3: Proposed binding of naphthoquinone derivatives (yellow; heme co-factor in green)
Figure 3: Proposed binding of naphthoquinone derivatives (yellow; heme co-factor in green)
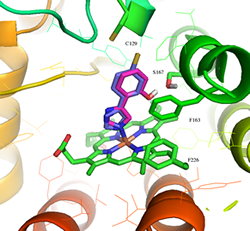
Figure 4: Proposed phenylimidazole derivatives (blue and purple) binding in IDO
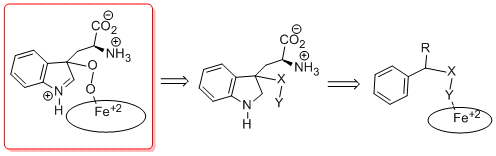
We are currently exploring a new structural class that is based on the reported mechanism of IDO’s catalytic reaction (Figure 5 and Syun-Ru Yeh et al. PNAS 2009, 106, 17371-17376).
Figure 5: Reported IDO mechanism
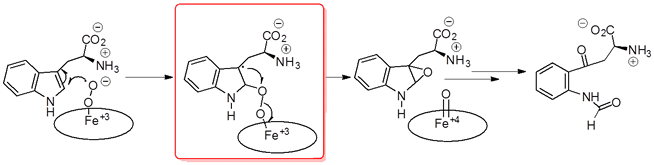 Mimics of the reported indole-peroxy intermediate shown in the red box are being generated and tested for selective and more potent IDO inhibition (Figure 6).
Mimics of the reported indole-peroxy intermediate shown in the red box are being generated and tested for selective and more potent IDO inhibition (Figure 6).
Figure 6: Current inhibitor design with X and Y being oxygen mimics.
We gratefully acknowledge support of this work from the National Institutes of Health (National Cancer Institute, R01 CA109542-04A2) as a collaborative project with biologists at Lankenau Institute of Medical Research.